
(I actually spent some time trying to confirm this, but without finding a reliable source.) The electrons kinetic energy is apparently converted into thermal energy (i.e. Each orbital has a capacity of two electrons. In the above orbital diagram, the box represents an orbital. Hence, draw the blank orbital diagram of silicon up to 3p subshell as follows: Blank orbital diagram of silicon. 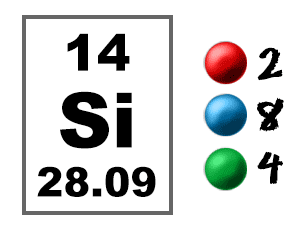
Report Error Is there an error in this question or solution Q 34. Using the same method, calcium has 2 valence electrons. ( 29 votes) Upvote Downvote Flag tyersome 6 years ago My understanding is that the potential energy is released as a photon. As mentioned above, the electron configuration of silicon is 1s 2 2s 2 2p 6 3s 2 3p 2. The electronic configuration of silicon is 2, 8, 4 and that of sulphur is 2, 8, 6. For the element aluminum, above, we see 2 electrons in the 3s orbital and 1 electron in the 3p orbital, so aluminum has a total of 3 valence electrons. These electrons are important because they are the ones that are gained, lost or shared in chemical reactions. We typically do not consider “d” electrons as valence electrons and therefore a more specific definition is needed: valence electrons are those electrons in the highest principal energy level. In many cases, “d” electrons will be present after the last noble gas, as in the element manganese : 4s 23d 5. The electrons written after the noble gas in brackets are called valence electrons. These electrons do not participate in chemical reactions. This list of electron configurations of elements contains all the elements in increasing order of atomic number. Those electrons in the brackets are called core electrons. The electron configuration shows the distribution of electrons into subshells. Valence Electrons-This notation for writing electron configurations helps us to highlight 2 different types of electrons in the atom. We may NOT use any element in the brackets, only noble gases. A schematic diagram of the designed all-silicon metasurface based on CP multiplexing encoding technique is shown in Figure 1, which can produce the off-axis tightly focused beams carrying specific polarization information in the focal plane 2830.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
